Tularemia is a uncommon however extremely infectious disease attributable to Francisella tularensis, a bacterium that may evade immune defenses. Symptoms of an infection can embrace fever, swollen lymph nodes and, in some circumstances, pneumonia.
What makes the pathogen particularly regarding is how little it takes to trigger an infection — fewer than 10 bacterial cells could be sufficient.
Scientists at Arizona State University have taken a key step towards understanding how this bacterium survives contained in the human physique. For the primary time, the group has remoted and studied a set of proteins that play a central function in an infection, revealing a potential weak spot that would ultimately be focused with new treatments.
The proteins sit throughout the bacterium’s inside membrane and seem to work collectively, forming small assemblies that assist it persist inside host cells. Until now, researchers had been unable to produce and stabilize these proteins within the lab, leaving a essential a part of the pathogen’s biology unexplored. By growing a way to extract and analyze them, the group has opened the door to extra detailed structural research and ultimately, new methods to disrupt the an infection course of.
The work builds on earlier studies of particular person proteins, however advances the analysis by capturing a gaggle of proteins that perform collectively as a system — one that’s important for an infection however had remained inaccessible.
“I am so excited about the project as we discover the structure and function of key proteins in the infection cycle,” says Petra Fromme, corresponding writer of the new examine. “By uncovering how these proteins come together and function, we’re starting to reveal a vulnerability in a pathogen that has been very difficult to target. This kind of insight is what lays the foundation for developing new therapeutic strategies.”
Fromme directs the Biodesign Center for Applied Structural Discovery and is joined by ASU colleagues, together with first writer Eranjalee Ranaweera.
The research seems in an advance on-line version of the journal Biochimica et Biophysica Acta (BBA)-Biomembranes.
Potent pathogen
Tularemia is a uncommon however doubtlessly severe disease attributable to the bacterium Francisella tularensis. People can turn into contaminated via insect bites — usually from ticks — in addition to contact with animals, contaminated meals or water, and even by inhaling airborne micro organism.
Once contained in the physique, the bacterium targets immune cells and might evade the physique’s defenses. Although circumstances are comparatively unusual within the United States, the disease could be extreme. Its ease of unfold and low infectious dose have led public well being companies to classify it as a high-priority pathogen. It has additionally been studied prior to now as a potential bioweapon.
Antibiotics can deal with tularemia, however rising resistance is a rising concern. Without immediate care, the disease could be deadly. Understanding how the bacterium infects and survives within the physique is subsequently essential for growing higher treatments and vaccines.
Unlocking a hidden system
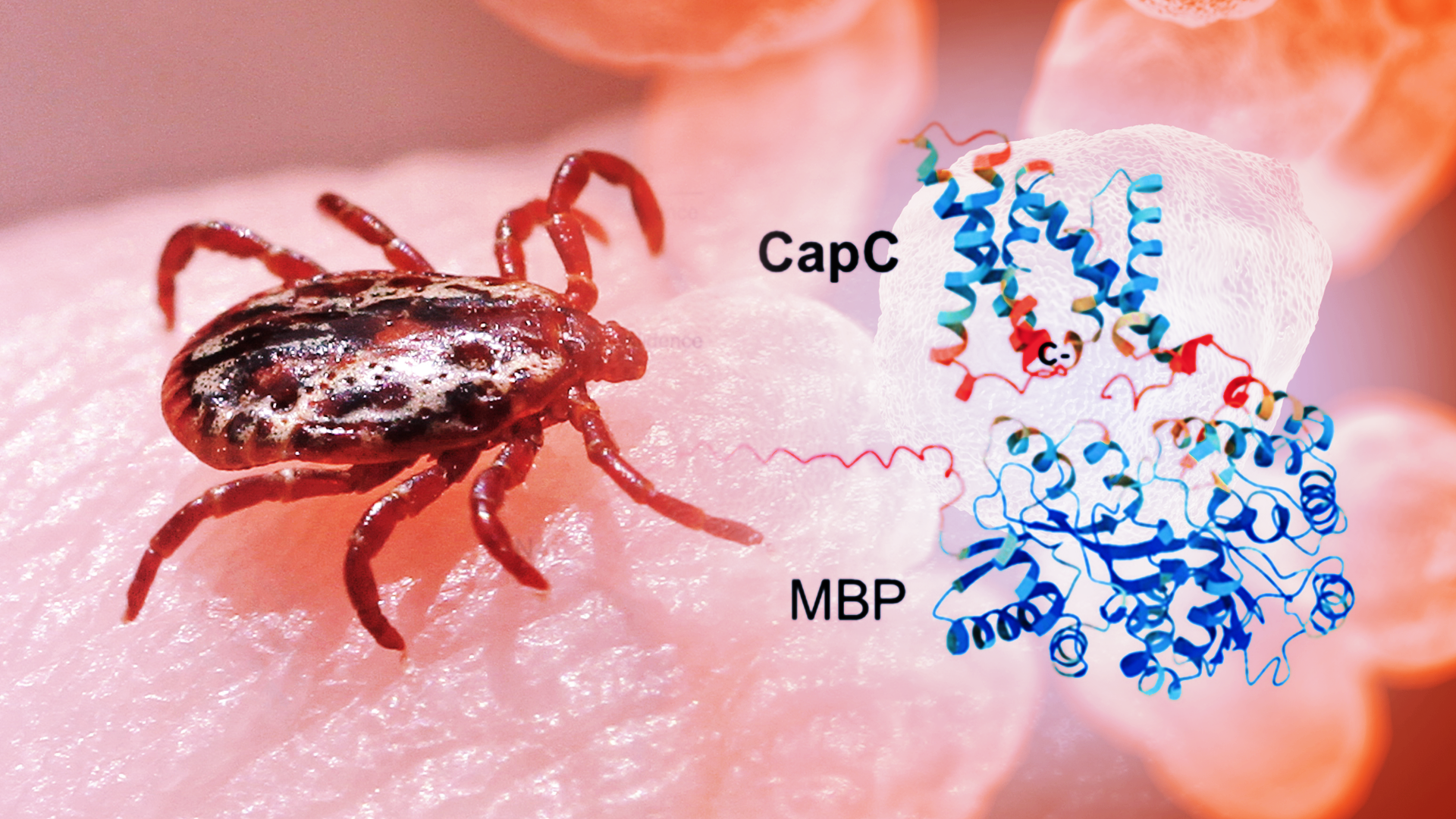
Researchers have lengthy identified {that a} group of proteins referred to as the CapBCA complicated is important for the bacterium’s means to infect and survive contained in the physique. These proteins are distinctive to F. tularensis, and animal research present that disabling them considerably weakens the bacterium. Thus, they’re a main goal for new therapy choices.
But focusing on these proteins has been tough. To design medication, scientists first want to perceive what these proteins seem like and the way they perform. Until now, that has been a serious problem as a result of the proteins are embedded in cell membranes, making them laborious to produce, isolate and examine within the lab.
To seize and examine these elusive proteins, the researchers discovered a method to produce them in laboratory micro organism. They inserted the related genes into the widespread bacterium E. coli. Then they added a molecular tag that helped information the proteins into cell membranes and made them simpler to detect and isolate.
Next, they used a detergent to gently extract the proteins with out destroying them. The proteins have been then remoted into clear, usable samples. This allowed the group to decide their form, how they assemble into small teams and the way secure they’re.
The proteins have been discovered to be principally spiral-shaped, sticking collectively into small teams, quite than performing alone. The group used a mix of imaging and biochemical methods to get the primary glimpse of those proteins, revealing their general form and the way they assemble, regardless that a full high-resolution construction remains to be to come.
“Isolating and characterizing these proteins for the first time gives us a way to visualize how the complex comes together,” Ranaweera says. “That’s a vital first step towards understanding its perform and figuring out it as a potential therapeutic goal, and it opens up many new instructions for future work.”
The analysis gives the primary structural clues to how this technique works, laying the groundwork for deeper examine and, ultimately, new therapies.